A biomedical startup is tuning electrical stimulation devices to your specifications so you won’t need pills.
Imagine, if you don’t experience it already, pain that doesn’t fade. Pain that throbs or burns, that keeps you on the couch, that ruins weddings and picnics and warm spring days. It might retreat to a dull ache from time to time, but the threat of resurgence is always there. It lurks. It changes you.
This pain resists aspirin or ibuprofen. It responds to more powerful opioids at first but gradually requires more and more pills to silence. Pills that are getting harder to get, because their addictive side effects have triggered an epidemic of overdoses that kill more than 30,000 people a year. Pills that, in any case, make you nauseated, lethargic, and constipated.
This is the kind of pain that Matthew Ward thinks he can fix — not with drugs, but with 50-year-old technology made new again. The key, he believes, is spinal cord stimulation, a method that uses implanted devices to jam pain signals with electric pulses.
Spinal cord stimulation, or SCS, has traditionally been a crude fix. The devices generally have had about a 50–50 shot at success. No one is entirely clear on why they work at all.
But Ward, a Purdue University biomedical engineer and founder of the startup Drug Free Therapeutix, believes he can make them work for a lot more people. Using modern-day software, Ward and his team are developing ways to make these old devices far more precise and reliable. Their hope is to make electricity, not drugs, the first line of defense against pain that won’t stop.
Hitting the sweet spot
Ward’s alternative painkiller builds on a discovery made in the 1960s, when pain researchers found that by using an implant to electrically stimulate the spinal cord, they could create a tingling numbness in place of throbbing and aching in the limbs and trunk. The first patient implanted with one of these “pain pacemakers” was a 70-year-old man with cancer in 1967.
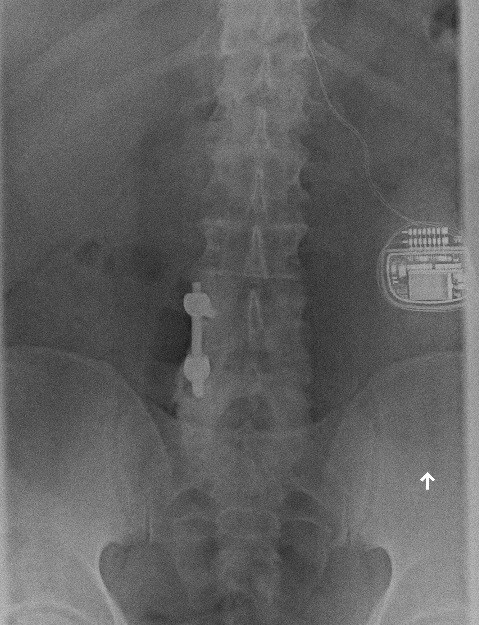
Today, around 30,000 people a year in the United States receive implants for pain. But the process—which involves implanting a power source roughly the size of a stopwatch in the abdomen and threading fine electrodes through vertebrae to the spinal cord—is a treatment of last resort. “Doctors typically won’t consider it until several first-line treatment options, like prescription opioids, fail,” Ward says.
This is partly because of the risks and costs of surgery. A 2009 study found that the cost to Medicare of implanting a spinal cord stimulator is almost $33,000, while private insurance can end up paying more than $50,000. Nonetheless, these devices are often worth the upfront outlay because they save on medical care in the long run, according to several other studies.
The real problem is that spinal cord stimulation hasn’t been very predictable. In one small study, for example, patients who had undergone back surgery and gotten no relief from their pain were given either SCS or another operation. SCS offered at least 50 percent pain relief in 47 percent of those who got the implants. That was significantly better than the 12 percent who received that level of relief from a second operation, but it was hardly a panacea.
That could change, though. Spinal cord stimulation has come a long way in the past decade, says Leonardo Kapural, a pain physician at the Carolinas Pain Institute and Center for Clinical Research. The batteries in the implants have become less bulky, and surgical techniques to anchor the electrodes in place have improved so that one common complication — electrode leads that move away from where they were implanted — has declined significantly. Researchers have also figured out how to better place the stimulators in the first place so they can hit “the sweet spot” for relief, Kapural says.
Even more promising are new trials using high-frequency stimulation, which block pain without creating the tingling feeling, known as “paresthesia,” as a replacement sensation. This kind of stimulation could open up SCS to new groups of patients, such as those with diabetic neuropathy, says Jijun Xu, an anesthesiologist at the Cleveland Clinic who is currently recruiting for a clinical study on the subject. People with this kind of pain from diabetes already have tingling, numb sensations in the extremities. High-frequency stimulation might improve their symptoms without simply piling on the tingling.
Kapural and his colleagues have found that with high-frequency stimulation, they can boost the percentage of patients who achieve at least 50 percent pain relief to 82 percent. That’s better than many pharmaceuticals on the market. The medication Lyrica, for example, helps just one out of about every seven patients who take it for nerve pain, Kapural points out.
Positive feedback
Ward’s technology is part of this new wave of SCS improvements. While doing his graduate work at Purdue’s Center for Implantable Devices, he was tasked with figuring out how a cranial nerve stimulator used to treat epilepsy and depression worked. This technology, known as neurostimulation, was once thought to block certain inputs, including ones that lead to pain, from traveling along the nerves. But the mechanism seems to be a lot more complex than that. More recent work suggests that stimulation releases pain-modulating neurotransmitters like gamma-amino butyric acid (GABA) and serotonin and soothes the sympathetic nervous system, which is responsible for stressed-out fight-or-flight responses.
Ward tried for a year to figure out why a given stimulus from the cranial nerve stimulator caused a given response, with little luck.
“I just could not replicate the results from any study,” he says. Finally, it dawned on him that looking at the stimulus as the starting point was a lost cause. What really mattered was the cranial nerve’s response and what that response ultimately changed in the brain. The same stimulus could cause vastly different responses in the nerve in different patients. Even the same patient could show variable nerve responses to the same stimulus over time. Doctors trying to calibrate pain-relieving implants for their patients are blind to this. “They just kind of crank up the current until it hurts,” Ward says. Then they “crank it down and then they leave it.”
Ward’s work revealed a way to get this crucial feedback. By modeling the nerve response to electrical pulses of varying strength and duration, Ward discovered he could predict the nerves’ responses to any given energy input. That led him to create a proprietary algorithm, which is what Drug Free Therapeutix is selling. Instead of tuning the pulses by guesswork, the company’s software adjusts continually to responses it measures from the nerves, in order to hit the notes that bring the patient relief. Now Ward’s company is working with device makers to add this software to neurostimulation units. Preclinical trials suggest they can boost the likelihood of pain relief from spinal cord stimulation from a flip of a coin to 80 or 90 percent.
Fewer pills
It’s not likely that spinal cord stimulation will push pharmaceuticals off the map. SCS wouldn’t be ideal for all types of pain. And there are low-tech alternatives for controlling pain, programs that include cognitive-behavioral therapy and physical therapy, that are well-supported by evidence. But patients are kept from these more complex treatments by uneven insurance coverage, according to a 2014 position statement from the American Academy of Pain Medicine. The result is that patients end up getting the easy treatment — often opioids — instead of the best one.
Still, Drug Free Therapeutix and other SCS improvements are among the very few new ideas that actually seem poised to make it to patients. Hurdles to developing new pain drugs are steep because pain is so perplexing. It’s not a simple stimulus and response, but a complex interaction between senses, emotion, and cognition. Chronic pain literally changes the brain, robbing regions that help integrate senses and emotions of gray matter. Pain can cause anxiety and depression, which can cause physical pain in their own right, making it hard to detangle which came first. Pain “is designed to be alerting and all-consuming,” says Barry Komisaruk, a researcher at Rutgers University who studies the basic neural pathways of pain. “It’s such a powerful stimulus, taking over the entire brain, that it’s very hard to control.”
Pain can be a side effect of spinal cord stimulation, too, a problem that Ward and his team hope their software can remedy. They also hope that making spinal-cord stimulation feasible for more patients—and pulling data out of more and more implants—will trigger innovations in device development. And that could bring relief to patients with nowhere else to turn.








