A first-person account of a simple clinical blood treatment that may extend healthy life.
Editor’s note: proto.life explores emerging medical technologies and the off-label use of existing treatments that may not have been clinically validated. We are always transparent about that when we do. In addition, sometimes our contributors have vested financial interests in their subject matter, which we will always disclose.
Watching blood flow out of my body—even within the sterile confines of a clear plastic hose—makes me anxious. I understand why: My predecessors in deep time who were comfortable with gushing wounds were naturally selected against. My anxiety is tempered by fascination. Staring at your blood is like leaning over a cliff. It’s the border between everything and nothing.
The ancient Greek philosopher-physician Hippocrates is best known for his maxim “first, do no harm” which did not exclude draining the blood of his patients. “What medicines do not heal, the lance will,” he wrote. Before Hippocrates, the Egyptians also practiced bloodletting, which remained a common medical therapy in many countries into the 19th century, before being thoroughly discredited.
Prematurely, as it turns out.
While it’s true that the draining of whole blood has little therapeutic value except in a very few diseases like hemochromatosis—aka “iron overload”—the latest scientific studies point to components of blood that accumulate with age and drive many different diseases—and potentially aging itself.
I’m sitting in a padded recliner in a nondescript room of a multi-tenant medical building in San Francisco. I have a view across a courtyard into what look to be upscale apartments, like a scene from Hitchcock’s Rear Window. My wife Song is in a similar recliner in a similar room next door. Each of us is attended by our own technician, overseen by Dobri Kiprov, the silverback MD who runs this “Apheresis Care Group.” The entire facility is immaculate and clutter-free, like Marie Kondo herself has already been here and gone home early.
“Apheresis” comes from the Greek, meaning, “to take away.” Clinically, apheresis involves drawing blood from the body and then separating it into components. The various cells are immediately returned to the body, while the liquid plasma is either processed further or discarded. Apheresis is how platelets and other blood products are extracted for donation.
Apheresis can also be used to remove from blood certain pathogenic (disease-causing) proteins—anything from LDL cholesterol to inflammatory molecules that drive sepsis to antibodies that cause autoimmune disorders. Apheresis is even being used experimentally to treat cancer by depleting tumor-generated immune inhibitors.

Practitioners of apheresis sometimes refer to it as a “subtractive” therapy—to highlight a key distinction from conventional drug therapies, which despite their many differences all involve the addition of molecules to the body.
Doctor Kiprov is one of the world’s foremost experts in therapeutic apheresis; he trained in clinical immunology at Massachusetts General Hospital and Harvard Medical School. Under his eye, my wife and I are undergoing a type of apheresis as part of a study on aging. There’s a well-reasoned hypothesis and growing evidence that removing inflammatory cytokines and other pathogenic proteins from blood could potentially rejuvenate tissues throughout our bodies. Though it remains to be proven conclusively in humans, Song and I see many potential benefits and no downside other than cost—so why not?
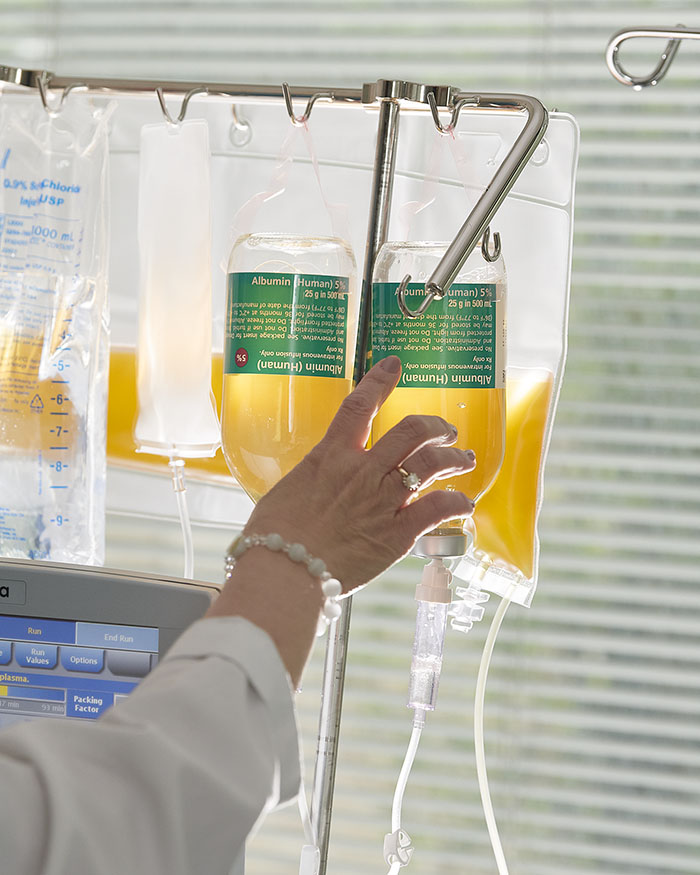
If you don’t like needles, be advised that the ones in my arms now are noticeably thicker than what’s used for vaccinations. My blood is flowing from my right arm through a tube into the dorm-fridge sized apheresis device, which hums and clicks as it separates my blood into cells and plasma—a yellowish, viscous liquid containing water, salts, enzymes, antibodies, and other proteins—including the problematic ones.
In this form of apheresis, my plasma—55 percent of my overall blood volume—is discarded. My blood cells are combined with a fresh mixture of 95 percent saline and 5 percent albumin—a soluble protein produced in the liver—plus a few “proprietary ingredients” that Kiprov declines to identify. My reconstituted blood is then returned to my body through my left arm. The process is called therapeutic plasma exchange (TPE) because my plasma is being exchanged for saline and albumin.
Both my palms are facing up as I watch my blood looping in tubes through the apheresis device and back into my body. I touch my thumbs to my middle fingers and feel for a moment like I’m meditating in the zendo of Roshi Frankenstein.
Could removing what ails you stop what ages you?
Cells communicate with each other via cell signals—molecules secreted by one cell and received by another. Signal molecules are too small to be seen even by electron microscopy so it’s not surprising that their discovery came more than two centuries after the discovery of cells. There are many types of cell signals that can trigger a broad range of cellular transformations. Some signals travel great distances—think dogs in heat—whereas others travel a micron or less—or even require cells to touch.
Immune cell signals are called cytokines. One type of cytokine that sparks special joy for scientists studying aging and disease are “death signals”—which as the name implies are cytokines that direct a cell to commit suicide. What sounds like extreme bullying in cellular junior high is in fact central to normal immune function. The immune system doesn’t fix anything directly. It distinguishes “self” from “non-self” and destroys the latter—if possible. “Non-self” includes cells transformed by viral infection, cancer, and mutation—as well as invaders like bacteria and parasites.
Death signals are normally delivered in a “paracrine” manner, meaning focally within a few microns of a bad cell cluster. This results in destruction of bad cells with minimal damage to normal ones. This is an evolved form of cell death known as apoptosis (from the Greek, “falling away,” like leaves from a tree). Apoptosis is akin to an implosion that contains viral or cancerous debris, the opposite of cell lysis—or rupture—which most viruses employ to spread their code
However, as we age, immune cells become less focal in delivery of cytotoxic cytokines, which gradually rise systemically, damaging tissue throughout the body—like the difference between pulling weeds by hand versus riding a mower across the flower beds while holding a tumbler of sangria.
Scientists focused on inflamm-aging believe that the gradual accumulation of inflammatory cytokines and related molecules has a profoundly negative impact on health.
While apoptosis always involves cell death, it manifests at the tissue level as inflammation. Whether inflammation is good or bad depends on which cells are dying. Inflammation of diseased tissue is good, while inflammation of healthy tissue—which happens more and more as we age—is bad.
Further aggravating the situation, some death signals are pleiotropic (from the Greek “many turns”). They’re cytotoxic to non-immune cells, but they recruit and activate immune cells, creating a feedback loop and contributing to “inflamm-aging.”
Scientists focused on inflamm-aging (myself included) believe that the gradual accumulation of inflammatory cytokines and related molecules has a profoundly negative impact on health—potentially accelerating aging as well. So the big question is: Would reducing the concentration of inflammatory cytokines in the bloodstream improve health outcomes?
There are already approved drugs designed to neutralize some of these cytokines. TNF inhibitors in particular are the largest drug class of all at $39 billion in annual sales; they include familiar blockbusters like Enbrel, Remicade, and Humira. These drugs can dramatically improve quality of life for people with autoimmune diseases, but they’re far from perfect. They commonly reduce immune competence, requiring warning labels that read like a sequel to the book of Job, warning of “serious infections leading to hospitalization or death… lymphoma and other malignancies, some fatal.”
The problem is that current TNF-inhibitors are not specific to the circulating, pathogenic form of TNF-α but also bind and neutralize the surface form of TNF-α on immune cells—where it’s mounted like quills on a porcupine, ready to fling at cellular foes. Ironically, the group that would most benefit from reduction of circulating inflammatory cytokines are the elderly, but they’re also the population least able to withstand a reduction of immune competence. Existing drugs are not a solution to the problem of inflamm-aging.
If only we could use a Star Trek transporter to beam inflammatory cytokines directly from our blood into outer space. Full disclosure: In 2016, with this in mind, I founded a company to develop a subtractive nanoparticle called a “NaNot,” capable of depleting specific pathogenic molecules—such as inflammatory cytokines—from the bloodstream without the toxicity of drugs. NaNots are essentially molecular apheresis, but they’re still a couple of years away from first-in-human trials. TPE on the other hand is a drug-free method of reducing inflammatory cytokines that is available now.
TPE is FDA-approved for a broad range of disorders, and is also being tested in clinical trials for treatment of COVID-19, idiopathic pulmonary fibrosis, and even cancers. The unifying hypothesis is that all these diseases are driven or enabled by factors in plasma, and thus that replacing patient plasma should improve health outcomes.
I don’t have any of the above diseases. But I do suffer, alongside all living beings, from aging. My parents both died of it, which was very unfortunate at the time, but now that I’m 60 and facing it myself, it’s a full-blown emergency demanding everyone’s best efforts. TPE may not extend my lifespan, but I’m intrigued by its potential for extending my healthspan—the number of healthy years I have left.
The end of the first treatment
After two hours, I’ve received six half-liter bottles of the saline–albumin solution. After the last bottle drains, the technician disconnects the needles from my arms, slaps on a couple of Band-Aids, and directs me to the conference room, where Doctor Kiprov and my wife are already waiting.
Kiprov must be in his 70s, given that his first scientific publication was 1975, but he looks fit and healthy—not surprising as I regularly see him zipping around our town north of the Golden Gate Bridge on his mountain bike. At work he’s old-school: white coat, suit and tie, lab reports neatly piled in front of him on the conference table. His voice is deep and tinged with a Bulgarian accent. He was born there and attended medical school in Sofia, the capital, before heading to the United States to further his studies at Harvard. In a later Zoom, I ask what led him to study medicine.

“I had no choice,” he says solemnly, with a hint of a smile. “My grandfather was a famous physician, and both my parents were physicians as well. In addition, Bulgaria was part of the Soviet Union then, and the government didn’t really respect or reward unskilled workers, at least not the way they rewarded physicians.”
I can’t resist: “Ironic that the worker’s party didn’t respect actual workers.”
Kiprov nods and replies, sotto voce, as if the secret police might still be listening, “Indeed.”
Prior to our TPE sessions, Song and I both had a full blood panel performed by a local lab, including measurements of our circulating inflammatory cytokines. According to Kiprov, my cholesterol and iron—which is proinflammatory—are both somewhat above optimum. Still, for a dude of 60, I’m abiding pretty well.
Despite being 12 years my junior, Song’s inflammatory cytokine levels are much higher than mine —especially her TNF-α, which at 14.7 pg/ml (picograms per milliliter) is more than twice the 7.2 pg/ml average for healthy individuals. This is more confirmatory than surprising. For the last decade Song has suffered recurring joint and tissue pain, ever since a bout of sepsis very nearly took her life. Sepsis is a phenomenon of escalating immune activation, a type of “cytokine storm” usually triggered by an acute infection that kickstarts widespread, runaway expression of TNF-α, IL-6, and related cytokines, leading to rapid destruction of organs throughout the body. Sepsis is the leading cause of death in U.S. hospitals and is arguably the greatest unmet medical need on the planet, killing more people annually than all forms of cancer combined.
People who survive sepsis often experience ongoing immune problems—either hypo–immune function (increased propensity toward infection) or hyper-immune function (increased propensity toward inflammation). The elderly often have both, as does Song prematurely.
Kiprov is optimistic about what TPE might do for us but emphasizes that lasting benefits could require six or more sessions. TPE is reimbursable for certain conditions—but not for aging, which the FDA does not recognize as a disease. The cost is significant—$6,000 per treatment, minus a discount because there are two of us—meaning that whatever systems might be rejuvenated by TPE, our savings will not be among them.
He sends us home, encouraging us to contact him if we experience any negative reactions. That evening and the next day, I don’t notice much of anything, but Song begins to feel different within hours. A decade of near-constant joint and tissue pain just… evaporates. Gone too are the blues that often discolor her evenings. Is her mood better because her pain is gone? Or is something different happening in her brain as well? That night she sleeps deeply and awakens at 7:00 am without any of her usual aches. She springs from bed and goes for a long walk with our (rather surprised) border collie, Foo-Man Chew.
My mind feels both still and ready, like a parked sports car.
I start noticing things myself the second night after treatment. As I lay my head down on the pillow, both my mind and body feel curiously quiet—no stray thoughts, no anxiety, no restlessness. My blood feels quiet. Was my blood noisy before? This sensation feels both new and oddly familiar. I realize that in my 20s and 30s I always felt this way at the end of the day. Describing the difference between then and now is like trying to describe being high or in love to someone who hasn’t experienced either—not that my post-TPE experience feels like being high or in love. Perhaps it just reminds me of those things because I first experienced them when I was young.
Light bulb! I feel… not exactly young, but younger. “Is this really possible?” I wonder, before slipping into a deep and peaceful sleep.
In the morning, I awaken with enthusiasm, like a child. My mind feels both still and ready, like a parked sports car. Song and I enjoy a relaxed breakfast before diving into work. Our next several days are rich in focus and largely devoid of stress. Song remains free of body aches, brain fog, allergies, migraines, and the other forms of suffering she regularly endures. I find myself taking dance breaks during the day—definitely “younger me” behavior. I also catch myself wiggling my fingers like a piano player sitting down to perform a concerto. I feel like I’ve been wearing gloves for the last decade and my hands are suddenly free.
Two weeks later
Precisely on the 14th day after treatment, Song wakes with a dull headache. Her seasonal allergies have returned. Her joint stiffness and pain begin to return that day as well, at a low level. A week later, Song has her first migraine since the treatment. Daily and especially at night, she again begins to experience a burning sensation in her joints and major muscles. She says her cognition feels slower and a familiar anxiousness has returned.
Both of us feel like we’re ahead of where we were before TPE—retaining perhaps 20 percent of the benefits we felt at the peak of improvement. A friend of ours—an investor in our company named Jim Winslow Foster, who also supports Kiprov’s research—went through six sessions of TPE, concluding in March of this year. He noted a diminution of benefit over time following each of his treatments, but less so with each one. The therapeutic hypothesis of TPE is that a series of treatments can potentially induce a reset of the immune system, leading to new homeostatic set-points—the values around which biomarkers normally fluctuate. Reducing the set points of inflammatory cytokines and other age-associated pathogenic factors potentially provides not just an immediate benefit but also remodels the organism as a whole, reducing secretion of these factors in the future.
It seems too good to be true, like magical thinking, but there’s no denying how TPE feels. Two weeks after treatment, Song and I are asking what this feeling means and how long it can possibly last. According to the second law of thermodynamics, entropy can only increase or remain constant in closed systems. The arrow of time can’t just reverse direction in mid-air.
Can it?
TPE in general and Kiprov’s program in particular is strongly informed by heterochronic parabiosis, from the Greek words “hetero” (other), “chronos” (time), “para” (next to), and “bios” (life). String it all together and you end up with an old mouse and a young mouse with their circulatory systems stitched together, like Siamese twins if they could somehow be born years apart.
Good God, why would anyone want to do this?
Parabiosis experiments, which have been done for more than 150 years, are an excellent way of identifying and studying various soluble factors in the blood of old versus young animals, as well as the relationship of these factors to multiple diseases. Some of the most significant parabiosis work—focused on the effect of blood factors on tissue and organismal aging—is by Irina and Michael Conboy, both faculty in the Department of Bioengineering at UC Berkeley. (Disclosure: some of the Conboy’s research is funded by a minor investor in my company.)
I arrange a Zoom with both of them. Irina is partially out of frame, typing furiously on her laptop. Michael is sitting calmly like a basketball player waiting for a game to start (he was a “nerd-athlete” in high school). I open with, “What’s it like to be married to each other and both be top scientists at the same institution in the same department?” Michael pauses before answering carefully, “As much as it drives you crazy, it keeps you from going insane.” If Michael is as skillful at developing anti-aging therapeutics as he is at answering loaded questions, we’ll all live forever.
How did both of them end up in the same field? “I have an unfettered view of science in general,” says Michael. “As long as I’m studying something and it’s new and interesting, I’m happy.”
“But Irina,” he says, glancing at her beside him on the couch, still typing away, “has always been interested in aging.”
I ask Irina what first triggered this interest. She replies in a crisp Russian accent that reminds me of music. “I became fascinated with aging when I was only five,” she says. “The difference between how I looked and how my grandmother looked motivated me to delay this change as long as possible—and ideally avoid it entirely!”

And what led them to investigate heterochronic parabiosis? “We were in grad school at Stanford, and had the idea that there was something (related to aging) in blood—because blood is common to all tissues,” Michael says. But initially they struggled with how to tease apart the various factors. “At Stanford there was someone doing parabiosis. We realized that this would achieve the same ends; it would test whether there was something going on in aging that was building up in blood.”
I ask what surprised them most about their first studies, published in 2005. “That the young mouse took a hit,” replied Michael. “Their youthfulness drops when parabiosed with an old mouse.”
Irina chimes in: “And we had seen it in cultured cells too, before that. When you put young stem cells into the old culture serum, they immediately age.”
The Conboy’s 2005 paper triggered tremendous excitement in the field of longevity science, especially their conclusion. “Our studies demonstrate that the decline of tissue regenerative potential with age can be reversed through the modulation of systemic factors.”
Many read the 2005 paper as evidence of some magical Ponce de Leon factor in young blood, despite the Conboys explicitly stating that the observed rejuvenation could be due to “an increase of positive factors in young mouse serum, a decrease or dilution of inhibitory factors present in old mouse serum, or both.”
That didn’t stop multiple companies from rushing to promote therapeutic applications of molecules highlighted in publications by the Conboys and their colleagues. Elevian for instance has raised more than $15 million to develop and test a therapeutic form of GDF-11—a growth factor with both positive and negative effects on the body depending on context. (Disclosure: my company has an investor in common with Elevian but no overlapping business activities.) Ambrosia began selling “young blood transfusions” in 2016 for $8,000 a pop before an FDA warning led the company to suspend operations in 2019. Silicon Valley is also rife with rumors of tech titans—excuse me for not sharing the names I’ve heard—arranging their own infusions of young plasma. (Unless modern youth are living substantially cleaner lives than my friends and I did back in the 80s, I think the FDA made the right call here.)
The Conboys realized that they needed to move beyond parabiosis, which involved too many variables to resolve competing explanations of results. “We needed to do a pure blood exchange,” recalls Michael.
So last year they devised a way to do just that by engineering a computer-controlled device for precisely exchanging blood between mice, without the mice being surgically connected to each other—an apparatus that closely mimics a human TPE setup. Experiments with this device revealed something profound: Virtually all of the benefits to the older mouse they observed in their original 2005 experiments could be replicated by mouse TPE.
“Our data demonstrate that a single (blood exchange) suffices to meet or exceed the rejuvenative effects of enhancing muscle repair, reducing liver adiposity and fibrosis, and increasing hippocampal neurogenesis in old mice, all the key outcomes seen after blood heterochronicity,” the paper concludes.
Last month, the Conboys, Kiprov, and their collaborators published again on the use of TPE, with new data from human studies—including the one in which Song and I are participating.
Aging is like a plane in a downward spiral, and we all see the ground rising beneath us.
In this paper, they describe aging as a “detrimental loop” in which protein dysregulation leads to cell and tissue damage, which in turn leads to the release of “pro-geronic” (pro-aging) molecules into circulation, which perpetuate and amplify the cycle. They present compelling mouse and human proteomic data suggesting that TPE mitigates three processes thought to drive aging: cellular senescence (loss of proliferative and functional capabilities); immunosenescence (age-associated decline of immune function in particular); and inflamm-aging, driven by the buildup of inflammatory cytokines.
The authors add that removing age-accumulated factors “abrogates their autoinduction,” which is science-speak for breaking the cycle of secretion of these toxic compounds, potentially remodeling overall biology toward a younger, healthier system.
Aging is like a plane in a downward spiral, and we all see the ground rising beneath us. Is TPE analogous to an airplane’s elevator, enabling us to level off and glide for awhile? Or is it more than that? Can TPE help us gain altitude? Much remains to be proven when it comes to the long-term benefits of TPE, but just as the first rule of flying is “don’t crash”, so too the first rule of living is “don’t die”—and staying healthier longer, which TPE seems to enable, supports that goal.
At the end of our Zoom, Michael summarizes the systemic effect of TPE by suggesting that it “looks like a reset or rebooting, like old computers.” And as if that analogy doesn’t date him sufficiently, he adds, “TPE is kind of like an oil change for your car.”
I’m not sure whether my body is really like a car or a computer or a spiraling airplane, but I like the idea that it’s fundamentally a machine, because even very old machines can continue to operate if properly maintained. I have no illusions that my machine will run forever, but thanks to the work of Doctor Kiprov and the Conboys, I’m optimistic I will at least outlast my warranty.
















