The ARDD meeting in Copenhagen this month featured new money, old experiments, endless enthusiasm, and a palpable sense of possibility.
Midway through the 9th Aging Research and Drug Development meeting (ARDD) at the University of Copenhagen earlier this month, a speaker stood up and shared some latest statistics from the U.S. Centers for Disease Control and Prevention (CDC). They showed another unsettling decline in American life expectancy—stark, even if not unexpected.
The longevity drop over the last three years has been a tale of two crises. The first, of course, is the worst pandemic we’ve seen in a century, with more than a million coronavirus-linked American deaths since its start. The second crisis and big driver of falling U.S. lifespans is drug overdoses, which reached all-time highs in 2020 and again in 2021. Overdoses now kill more than 100,000 Americans a year.
U.S. life expectancy declines are also a tale of two countries—or one single country suffering widespread health disparity. As just one example, last year’s lifespans fell the furthest for Native Americans, erasing 40 years of longevity gains.
All that is a stern reminder of why this emerging, cross-disciplinary field of longevity matters—and why proto.life came to Copenhagen for what many consider to be the top scientific meeting in the field. The decline in American life expectancy says it all: Human lives are at stake, and health disparities must fall.
Longevity is not only about one single disease, or even many diseases. Nor is it only about piling on a few extra years of life. It’s not a field marked solely by flashy tech, ambitious startups, and unproven treatments. ARDD showcased one medical leader after another who spoke of safety first, signaled a selfless devotion to the science, and lauded the field’s its potential to be a democratizing force in the future of medicine.
“Life is such a beautiful thing, and we need to make it available for everybody without disease,” meeting co-organizer Alex Zhavoronkov said in the opening session. “And that’s where longevity medicine comes into play.”
Here are seven lessons I took away from ARDD 2022.

Lesson 1: Primum non nocere
Longevity is not what it used to be. According to some, the field is at an inflection point: moving from expanding to exploding. Our body of knowledge is growing, and with it, so is the community. There are more startups, more university faculty, more hospital clinics, more students, and more research papers—so much so that people at the meeting were already starting to reminisce fondly of those first ARDD gatherings just a few years ago when only 50 people showed up. (According to Zhavoronkov, this year’s hybrid meeting had more than 400 in attendance and thousands more joining online.)
One sure sign of the field’s growth was the announcement made on the conference’s first day about the August launch of a new organization called the Longevity Medicine Society. It’s a concrete step toward enshrining longevity as a bona fide discipline, and it will also shake up the field.
“Longevity medicine is crystallizing as a discipline.”
“The idea was to build a society like we have for all the other disciplines in medicine,” says Evelyne Bischof, a professor of medicine at Shanghai University of Medicine and Health Sciences and the inaugural vice president of the society. She has previously spearheaded educational efforts with Zhavoronkov and others, co-developing a formal course on longevity medicine for doctors. At the ARDD meeting, Bischof announced their course had just received continuing medical education (CME) accreditation from the American Medical Association.
“Longevity medicine is crystallizing as a discipline,” says Andrea Maier, an internal medicine specialist and geriatrician at National University of Singapore who is serving as the society’s inaugural president. One thing that’s not yet clear, several experts told me, is whether longevity will come to be established as a sub-discipline of geriatrics or internal medicine or whether it will become a separate medical specialty unto itself.
“Whichever way it goes,” Maier says, “it’s happening.”
The need for all this organizational development is not simply a matter of bookkeeping. Defining longevity as a bona fide medical discipline will help doctors help their patients. The first maxim of all doctors is: Do no harm, but Primum non nocere never assumes you have to go it alone.
“We need guidelines,” Maier says.
“At this stage,” says Gianfranco Vettorello, an Italian vascular surgeon I met at the meeting, “it is difficult to have guidelines. We don’t have a specialty.” Without a specialty, there is no designated authority. With no such authority, there are no official guidelines. Without guidelines, doctors may not have the tools they need to weigh the best options for their patients. Vettorello is very familiar with that problem from his practice. “In Italy,” he says, speaking generally of the entire profession, “medical doctors know nothing about longevity.”
That’s ironic, he adds, because Italy is a brand—an eating brand, a lifestyle brand, and because of the long lifespan “blue zone” in Sardinia, it’s a longevity brand. There’s credibility in the Italian lifestyle, Vettorello says, which is why he started a separate organization called the Italian Longevity League—to translate evidence-based medicine and educate doctors as well as professionals outside the clinic, including engineers, architects, geologists, and restaurateurs. They want to carefully weigh the evidence-based components of the Italian lifestyle that, along with other interventions like drugs, can be translated into practice.
“We need bigger clinical trials to really evaluate in phase 3 and 4 studies if certain supplements or lifestyle interventions or repurposed drugs are going to work. And we need to evaluate the effect size of these interventions,” Maier says. “We need cross-country randomized controlled trials with different ethnicities, different cultures—because environment might be an effect modifier in what we are finding in terms of the effect of the interventions we are doing.”
“We need that evidence,” she says.
Guidelines can shape outcomes for whole hospital clinics, not just individual practices. Earlier this month the National University of Singapore officially launched its Center for Health and Longevity, which Maier co-directs. She says the Longevity Medicine Society wants to create frameworks or blueprints for new longevity clinics—including those at publicly funded hospitals like hers.

Lesson 2: Stare into the sun
I am sitting outside the Great Hall at the University of Copenhagen at a small outdoor café on the cobblestone street that served as my de facto office during the conference. Today I’m meeting with Anastasia Georgievskaya, the CEO of the Estonia-based company Haut.ai, which uses AI to develop skin care assessment software for clients at longevity companies and in the cosmetics industry (haut is the German word for “skin”).
We met so that she could demo her new app. Founded in 2018, her company now has 26 employees, mostly engineers. They develop software-as-a-service tools to help client companies do things like market research, product refinement, and post-market analysis. They quantitatively and qualitatively analyze a person’s skin in an unbiased way, she claims.
“Every person is really biased,” Georgievskaya says. “So we exclude the bias component.”
It’s hard to be objective and unbiased when looking in a mirror or a selfie. We underestimate the quality of our skin, focusing on every clogged pore, each minor blemish, or any tiny wrinkle. We find flaws in our faces nobody else would see, we think we look older than we do, we imagine our faces falling short even if our skin looks fine, and we turn away from the mirror or photo in horror and disgust.
AI is the solution to that, Georgievskaya says, and we’re here at the café so she can show me how it works.
She has me staring almost straight into the sun. It’s blinding, and I can barely keep my eyes out of squint mode. Will that affect the image quality, I ask. No, she says. So I tilt my head up more and try to hold still long enough for the software to be satisfied. The app tells me to position the phone even closer. It’s inches away from my face, finally, before the software seems satisfied. The camera snaps. The shot uploads. And we sit down to finish our chat. Somewhere in Estonia, I imagine, an AI server and a bunch of 20-something engineers are busy processing images of my face… Oh God!
What the technology does, Georgievskaya says, is to measure and assess a person’s skin in a reproducible way—the same objectively analysis today, tomorrow, in a week, or in a month—based on proprietary biomarkers her company has developed. That gives them the ability to assess an individual’s need for skin care products or assess the impact of one product over another.
“This is exactly what your skin condition is right now,” she says, once the report uploads.
The results? Somewhat mixed. The AI guesses my age as a few years younger than I am, so that feels good. I score an exhilarating 100 percent, the highest ranking, on the acne index, and my pigmentation score is also excellent. But the report goes rapidly downhill from there. My pore health is somewhat ho-hum. My wrinkly line score is only average. My skin redness and translucency are both bad. And the AI says my skin hydration is horrible. Yikes!
“This is what our technology does,” Georgievskaya says. “We tell you this is exactly what your skin condition is right now.”
That’s fine with me, I think—but can we talk about my excellent pigmentation again?
Lesson 3: Safeguard the data
Trusting the data was actually one of the big themes of the meeting, and we heard over and over how biomedical research is really starting to embrace big data and artificial intelligence.
But it’s not enough to design an AI to analyze huge amounts of data—your study also needs to comply with HIPAA and other privacy laws, which often makes collaborations that extend across departments and institutions more difficult to do. Some experts at the meeting touted the power of an approach called “federated learning” as a solution—a decentralized machine learning approach where separate users or sites collaboratively train AI models run by a central server. The models cross institutional boundaries, but the data stays local.
“Basically, it allows you to completely anonymize the data,” Zhavoronkov says. In addition to being a co-organizer of the meeting, he is also the founder and CEO of the Hong Kong-based biotechnology company Insilico Medicine, which develops AI and robotics platforms for drug discovery, among other things.
Lesson 4: Follow the money
Let’s get down to one of the central questions of longevity medicine: When will we have approved drugs on the market to increase a person’s life span, health span (healthy years of life) or both? One expert told me that at the moment we have lots of longevity drugs that look really promising in worms, a few things that are promising in mice, and nothing at all approved by the FDA for use in humans.
For every drug that pharma gets approved, biotechs get eight approved for the same amount of money.
Nobody seems to doubt that will change, and more than one expert predicted it will happen soon. One speaker at ARDD even stood and vowed, like a biotech equivalent of Joe Namath, that his company would have a drug on the market within a decade. “I guarantee a victory,” Namath famously told reporters the night before his underdog New York Jets won Super Bowl III over the Baltimore Colts. The media and fans loved it, even if his coaches cringed.
Part of the reason for the tangible exuberance in longevity today is there are new opportunities beginning to emerge—especially for biotech startups. “Pharma is in a sort of crisis,” says Sebastian Brunemeier, the co-founder and CEO of ImmuneAGE Pharma. “Some people think they picked the low-hanging fruit already.” He points to data from the NIH showing that for every drug that pharma gets approved, biotechs get eight approved for the same amount of money.
“It’s created this huge void, this gap in the market for companies doing preclinical and early clinical R&D,” he says.
Confidence is also peaking because of an influx in new funding. The triple-unicorn biotech company Altos Labs officially launched in January this year to great fanfare over its luminary investors, stellar science advisors, and $3 billion initial fundraising.
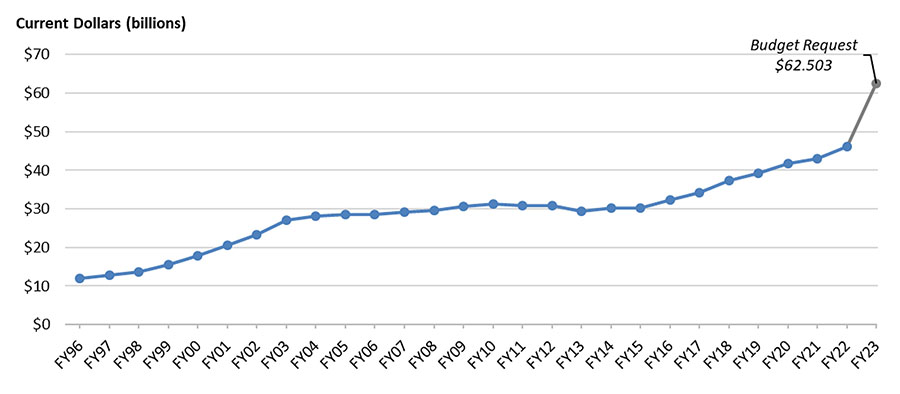
Public funding also looks promising. President Biden’s proposed FY2023 budget includes a whopping 35.3 percent increase for the overall National Institutes of Health (NIH) budget—though it remains to be seen how much Congress will actually appropriate of the requested funds. The FY2023 proposal brings NIH discretionary budget authority to $49 million, continuing an almost 10-year trend of steady increases. It also proposes increasing investment in the Advanced Research Projects Agency for Health (ARPA-H) to $5 billion (it’s currently at $1 billion), which is significant for the field because that agency’s initial mandate is to focus on the massively aging-relevant health problems of cancer, diabetes, and dementia.
There are also hints that at least one big pharma company is poised to throw its weight into longevity. One of the unverified tips I heard at the meeting is that New York City-based pharma giant Pfizer will soon be announcing their entry into aging research. No doubt others will follow.
But very few splashes in science make as many ripples as the promise of new funding that came earlier this year with the announcement of the launch of the Hevolution Foundation, a nonprofit organization established by royal decree at the behest of Saudi Prince Mohammed Bin Salman, which is looking to spend $1 billion per year on longevity.
All eyes were on Hevolution’s two speakers at the ARDD meeting, including CEO Mehmood Khan, who announced at the meeting Hevolution’s initial funding call, a partnership with the American Federation for Aging Research (AFAR) to administer a number of $375,000 junior investigator awards and a separate fund for sponsoring scientific conferences to “foster scientific exchange, networking, and collaboration.”
“This is the first of what we see as a series of future collaborations,” Khan says. “There’ll be many more in our mind.”
In his talk and in a separate one-on-one interview later in the week, Khan gave few specific details on what sort of other projects they will fund. But what he said hints at a planned portfolio of large and small grants funding both early-stage and late-stage research. They now appear to be developing a portfolio strategy—“a logical portfolio strategy,” Khan says.
Lesson 5: What do the tea leaves say?
Again, the details are scarce, but here’s the rough sketch. What proto.life took away from the ARDD meeting hints at a funding strategy whereby Hevolution would approach the world of longevity research through at least four different lenses—research stage, career stage, disciplinary field, and geography. And there was a strong sense they will seek to appropriate funds across the spectrum of each.
- The AFAR announcement was related to developing junior investigators, and if we had to guess, we would predict similar partnership vehicles in the future for funding investigators with larger, more established labs. But for now, the only thing we know for sure Hevolution is funding (other than conference funds) is focused on future, upcoming science leaders. “I told the team: Be very careful it’s not just a repackaged senior investigator putting the name on of a junior—we really want to develop these juniors.”
- We surmise they plan to fund both late stage projects devoted to translational research as well as early career and early stage projects. Though the CEO offered no specifics on which late stage projects they will fund or when, he did offer a hint that they could be funding projects at the earliest end of the stage-of-reseach spectrum—those most initial ideas, where there are concept hypotheses but no pilot data. “There’s a need for that,” Khan says. “NIH won’t fund that.”
- Khan also says they want to make sure their portfolio includes projects and experts from disciplines not traditionally represented in these fields.
- Finally, Khan says he instructed his team to ask whether they are bringing in the best minds regardless of geography, including people from countries or institutions that may not have the best track record for doing successful science. “That’s okay,” he says. “Aging is a global issue.”
Institutional background geodiversity is not something traditional university departments have been terribly great at achieving. Just this week, a paper in Nature showed that the vast majority of U.S. faculty members (295,089 people hired in 10,612 departments from 2011–2020) received doctorates from only a small number of schools. It’s another instance of the 80/20 rule: 80 percent of faculty members trained in the United States received their PhDs from just 20 percent of prestigious U.S. universities.

There were three other big takeaways from the interview worth sharing. One was Hevolution’s stated commitment to developing new tools that will benefit the entire field. The second was a sense that they are not going to wait to develop new drugs. Khan acknowledged to proto.life how difficult, time consuming, and expensive it is to develop drugs for any indication—let alone aging—but he said the solution should never be to wait. We could spend the next ten years talking about how difficult it is or we get going, he says. “Let’s start—that’s the first thing.”
The third takeaway harkens back to the reality enshrined by the falling U.S. life expectancy—a reminder that longevity, like any other field of medicine, is one where human lives are at stake and health disparities are rampant. The vision Khan articulated for Hevolution was above all a democratizing one, centered around expanding health for the benefit of all and doing so as a public good.
“One hundred percent of our profits from our venture fund will go back into the charity,” Khan says. “We’re not doing this for financial return.”
Lesson 6: Listen to your elders
Like all of modern medicine, longevity starts with observations, proceeds to rich data, develops a deeper, mechanistic understanding of nature, translates that understanding into safe, clinically tested interventions, and seeks to take them to the marketplace and improve health outcomes for people everywhere.
Some of the my favorite observations discussed at the ARDD meeting come from studies involving centenarians, people born in or around World War I who lived past 100. A number of cohorts even include “supercentenarians,” who are alive past 110. What their stories can tell us about aging may surprise you.
Kaare Christensen is a principal investigator on the National Institute on Aging-funded Long Life Family Study, which follows a cohort of some 5,000 people from hundreds of families in the United States and Denmark who have siblings with exceptional longevity. The study shows that when they, their children, or their grandchildren get diseases, their survival tends to be better than age-matched controls who are not from such families.
“All three generations have better survival,” Christensen said during his talk at the meeting. “It’s clear they get fewer diseases, but it’s not specific diseases.” There are no cancer-avoiding families or heart disease-avoiding families, he said. Instead, the data shows that the centenarians, their children, and their grandchildren are generally good at avoiding all sorts of health threats.
Nir Barzilai, a professor at the Albert Einstein College of Medicine of Yeshiva University in New York also spoke about a large cohort, which includes about 3,000 people of Ashkenazi Jewish descent, of whom 750 are centenarians. They include four siblings who were born in Europe between 1910–1920. He was surprised by one of these centenarians when he went to meet her one time more than a decade ago. She was 100 at the time, and Barzilai was floored when she answered her door smoking a cigarette. “I asked her, ‘Tell me, none of the doctors told you to stop smoking?’ And she said, ‘All four doctors who told me to stop smoking, they died,’” Barzilai said in his talk.
As humorous as that anecdote is, it hints at one of the big lessons centenarians could teach us: what aspects of geroprotection is genetic. “[The offspring of centenarians] have half the mortality, half the cardiovascular disease, half the Alzheimer’s,” Barzilai said. “It’s really quite incredible.”
The mechanism may turn out to be complicated. The situation is surely not as simple as a single gene conferring longevity. Exceptional longevity is a complex trait, and it’s likely to involve many different genes as well as significant “nurture” components, like contributions from the environment, from diet, from exercise, and from other lifestyle choices as well. Even so, what’s really compelling about studies involving long-lived humans (or other creatures) is the idea that understanding these genetic mechanisms could lead to new drug targets.
That’s the exact idea, says Kristen Fortney, the CEO of BioAge, a biotech startup in Richmond, California, which she describes as “a clinical stage company advancing an internal portfolio of therapies that treat disease by targeting aging.” Targeting the mechanisms of aging starts with identifying those targets, she says, and that’s where the centenarian research really shines.
“We’ve partnered with some really special biobanks that started collecting blood samples from healthy, middle-aged people 50 years ago,” Fortney says.

Through the years, the biobanks collected blood from the subjects as well as clinical health data at every annual exam. So they have rich clinical data on things like cognitive function, walking speed, and grip strength. What Fortney’s company has done is to molecularly reanalyze those blood samples, which in many cases have been locked away in freezers for decades and look at the thousands and thousands of proteins and metabolites they contain.
“[It’s] like a 50-year experiment, basically,” she says. That allows them to reconcile the molecular data with clinical observations and ask what is different in the blood of a 50-year-old who dies at 60 versus someone born in the same year who makes it to 100.
“That’s where our hypotheses come from,” she says. “That’s where our targets come from.”
In the last few years, BioAge licensed the drug BGE-105 (originally called AMG 986) from biotech giant Amgen in Thousand Oaks, California. That drug is in phase 1 clinical trials for combating age-related muscle loss. They also licensed the drug BGE-175 (also known as asapiprant) from Osaka, Japan-based pharmaceutical giant Shionogi & Company. They are now testing it in phase 2 clinical trials for the treatment of COVID-19 and influenza, where outcomes are made worse by age-related immune function declines. BioAge has a third drug in phase 2 trials for unexplained anemia of aging, which is called BGE-117 (licensed from Tokyo-based Taisho Pharmaceutical Company). Fortney says they also have a fourth drug in the pipeline, their first designed entirely in-house, which is coming along. They are on track to file an investigational new drug application with the FDA next year.
“And then we move into clinical trials,” she says.
In a very colorful metaphor, zoologist Emma Teeling of University College Dublin likens the immunological aspects of a centenarian’s biology to a Ferrari with really good brakes—a car that’s fast as hell but can stop on a dime without setting the tires on fire.
“When you look at supercentenarians and you look at their immune response, what you find is they are really, really inflamed,” she says. However, they have an equal amount of anti-inflammatory markers to maintain this homeostasis. That means they mount enough of an immune response to fight off infections but match that with enough of an anti-inflammatory response to avoid an autoimmune shock, she explains. And then she leans in over the table and lowers her voice for dramatic effect.
“The bats do this too,” she says.
Lesson 7: Pay attention to bats
For the past 25 years, Teeling has been looking at the biological history of bats, exploring their genes and genomes and the evolutionary changes that allowed them to evolve things like sound echolocation and winged flight, an ability unique among mammals. They are also one of the most successful class of species in Earth’s history—about a fifth of all the mammals on the planet are bats. But there’s another intriguing thing about bats.
“There are 19 species of mammal that live longer than humans, given their body size. And 18 of these are bats.”
“They are the longest lived—given their body size—of all mammals,” Teeling says.
Nature’s general rule of thumb is that small things age fast and die young whereas big things age slowly and live longer. Compare your own expected lifespan to that of an average mouse (which may live a few years at most) versus a mighty, 13-foot bowhead whale (which can easily live to 200). Bats buck that trend.
“There are 19 species of mammal that live longer than humans, given their body size,” Teeling says. “And 18 of these are bats.” (The 19th species is the naked mole rat.)
Bats appear to have evolved myriad genetic ways to slow down aging. Pathological exams of some of the longest-lived bats show that they do not appear to get cancer. They are also known to tolerate and live with many different viruses and pathogens (coronavirus, anyone?).
For the last 10 years Teeling has led a group of researchers across the English Channel every year. They ferry a mobile lab to the north of France, where they drive through the pastoral hills and picturesque towns of Brittany, arriving at an old Gothic church whose belfry serves as both flophouse and nursery to a population of around 5,000 bats. Every year she and her colleagues get together with local conservationists and townsfolk to capture, weigh, measure, and release every bat in this population, repeating the same procedure again with the same bats the next year.
“We have this fantastic dataset,” Teeling says. “And we can now ask the question: What happens in bats as they age?”
They have looked at the length of the bats’ telomeres—the protective caps on the ends of DNA chromosomes that shorten every time a cell replicates. They found that in some species of bat, the telomeres do not shorten. Bats also have an incredibly fast metabolism—something hardly surprising for a creature who spends a great deal of its life (and energy) staying aloft. But a high metabolic rate would sometimes be expected to cause oxidative damage to cell mitochondria, but they have looked at bat cell mitochondria and they don’t see it. “Which means they have ways to either repair their mitochondrial damage or remove it,” she says.
Looking at the bat genome and transcriptome, they found that as bats age, they increase their ability to repair their DNA. There is also an expansion of antiviral genes along with the loss of some inflammasome genes (think Ferrari with good brakes). “You’ve got to be able to get your immune response to be appropriate enough to neutralize a pathogen but then not make you sick, and the bats seem to develop a sweet spot between the two,” Teeling says. “That’s why I study them.”
In an interview with proto.life, Teeling described a pathway from basic discoveries of targets in bats to translation of those targets in preclinical mouse studies and then into clinical trials in humans—maybe.
“A perfect world would be stuff that we found in the wild that we were able to directly translate that made differences to human lives,” Teeling says. “That would be perfect.”
So that’s it. To reiterate and expand on the seventh longevity lesson from the meeting: Pay attention to bat studies. They’re not just for COVID-19 anymore.
















